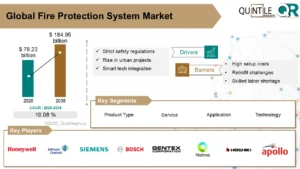
GSK, Pfizer, and Sanofi have been granted relief after a court ruled that claims of Zantac-linked cancer were not “supported by science.”

GSK Plc, Pfizer Inc, Sanofi SA, and Boehringer Ingelheim were released from thousands of U.S. lawsuits alleging that the heartburn medication Zantac caused cancer on Tuesday after a judge ruled that the claims were not supported by sound science.
GSK Plc, Pfizer Inc, Sanofi SA, and Boehringer Ingelheim were released from thousands of U.S. lawsuits alleging that the heartburn medication Zantac caused cancer on Tuesday after a judge ruled that the claims were not supported by sound science.
According to a report by US District Judge Robin Rosenberg in West Palm Beach, Florida, the ruling dismisses approximately 50,000 claims in federal court.
However, it has no bearing on the tens of thousands of similar cases currently pending in state courts across the country.
“We are extremely surprised by this miscarriage of justice,” said the plaintiffs’ lawyers in a joint statement, and “fully expect” the ruling to be reversed on appeal.
According to a Sanofi spokesperson, the decision “significantly reduces the scope of the litigation potentially by more than 50%,” with the remaining litigation taking place only in state court. Furthermore, a spokesperson for GSK stated that the company welcomed the decision, and Pfizer stated that it was pleased with the outcome.


In a statement, privately held German drugmaker Boehringer said it was looking forward to “continuing our vigorous defence of the remaining cases in state courts.”
According to Reuters, all of the drug companies have denied that Zantac causes cancer.
Zantac, which was approved in 1983, became the world’s best-selling medicine in 1988 and one of the first drugs to sell for more than $1 billion per year. Initially marketed by a predecessor of GSK, it was later sold to Pfizer, Boehringer Ingelheim, and finally Sanofi. Numerous generic drugmakers also released versions of the medication, but none are named as defendants in federal court cases.
Some manufacturers and pharmacies stopped selling the drug in 2019 due to concerns that its active ingredient, ranitidine, degraded over time to form a chemical known as NDMA. While NDMA is found in trace amounts in food and water, it is known to cause cancer in higher concentrations.
The U.S. Food and Drug Administration removed all remaining brand names Zantac and generic versions from the market in 2020, citing research indicating that the amount of NDMA in the products increases with storage time and could potentially become unsafe.
According to reports, lawsuits from people who claimed to have developed cancer after taking Zantac began to pile up soon after the recalls began. Plaintiffs claimed that the companies were aware, or should have been aware, that ranitidine posed a cancer risk and failed to warn consumers.
The lawsuit initially included claims of over ten different types of cancer allegedly linked to the medication, but it was later reduced to five: bladder, esophageal, gastrointestinal, liver, and pancreatic cancer. Some claims involving other cancers are still being heard in state courts.
You May Be Also Interested in:
- Covid-19 was a ‘man-made’ virus: According to a scientist who worked at the Wuhan lab
- The G20 presidency of India will be a “watershed moment” in its history: Ambassador Ruchira Kamboj
- World Powers Extend Support as India Takes Over the G20 Presidency
- Elon Musk reduces employee benefits at Twitter and eliminates several perks in order to increase profits
- Ben Simmons of the Nets was booed a lot by 76ers supporters in his first game back in Philadelphia
- Closest Moon Fly-by by NASA’s Artemis 1 Orion Spacecraft Ends with Stunning Images