Live Biotherapeutic Products and Microbiome CDMO Market Outlook 2035: Growth Drivers, Trends, and Strategic Insights
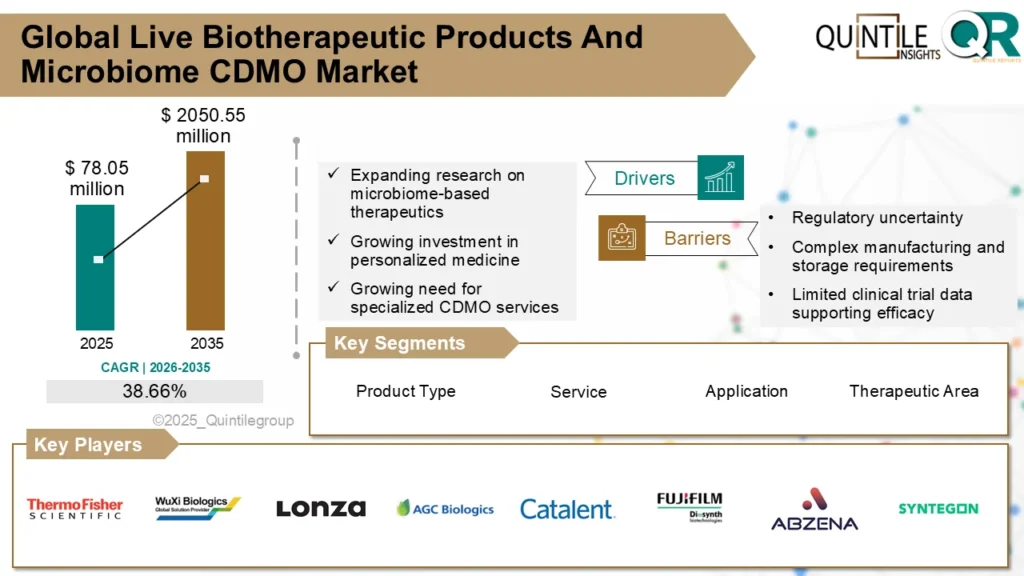
The Live Biotherapeutic Products and Microbiome CDMO Market is entering a transformative growth cycle as microbiome-based therapeutics transition from exploratory research to regulated pharmaceutical commercialization. With expanding clinical pipelines, increasing regulatory clarity, and rising demand for specialized biologics manufacturing, the Live Biotherapeutic Products and Microbiome CDMO Market is poised for sustained expansion through 2035.
For biotechnology companies, pharmaceutical leaders, contract manufacturers, and institutional investors, capabilities within the Live Biotherapeutic Products and Microbiome CDMO Market are rapidly becoming a strategic differentiator in advanced therapeutics manufacturing.
Free Sample Report :- https://www.quintilereports.com/request-sample/1110-live-biotherapeutic-products-and-microbiome-cdmo-market/
Market Overview: Evolution of the Live Biotherapeutic Products and Microbiome CDMO Market
The Live Biotherapeutic Products and Microbiome CDMO Market is witnessing accelerated global adoption as pharmaceutical developers increasingly outsource complex microbial manufacturing. As regulatory pathways mature and clinical pipelines expand, the Live Biotherapeutic Products and Microbiome CDMO Market is evolving into a high-growth segment within advanced biologics and specialty fermentation services.
Live Biotherapeutic Products (LBPs) require:
- Structured clinical trials
- Regulatory approval processes
- GMP-compliant manufacturing
- Strict quality and safety validation
Unlike traditional probiotics, these therapies demand pharmaceutical-grade development and validated microbial production processes.
As microbiome therapeutics advance across gastrointestinal, metabolic, immunological, and oncology indications, demand for specialized Contract Development and Manufacturing Organizations (CDMOs) continues to accelerate globally.
Manufacturing within the Live Biotherapeutic Products and Microbiome CDMO Market presents unique complexities, including anaerobic fermentation, strain stability management, and contamination control — capabilities requiring advanced infrastructure and regulatory-aligned facilities.
Key Growth Drivers in the Live Biotherapeutic Products and Microbiome CDMO Market
1. Expanding Clinical Pipeline for Microbiome Therapeutics
The global pipeline of microbiome-based therapeutics has expanded significantly over the past decade. As late-stage clinical programs progress toward commercialization, the Live Biotherapeutic Products and Microbiome CDMO Market is experiencing rising demand for scalable, validated, and compliant production capacity.
Therapeutic focus areas include:
- Recurrent gastrointestinal infections
- Inflammatory bowel disorders
- Oncology adjunct therapies
- Metabolic diseases
- Immune-modulating treatments
Commercial-scale readiness is becoming essential as regulatory submissions increase.
2. Regulatory Framework Evolution Supporting Market Expansion
Global regulatory authorities have established clearer guidelines for live biotherapeutic products, reducing ambiguity around classification, safety standards, and microbial quality control requirements.
Improved regulatory clarity strengthens:
- Investor confidence
- Cross-border partnership activity
- Long-term manufacturing planning
CDMOs operating within the Live Biotherapeutic Products and Microbiome CDMO Market are aligning facilities and validation processes with evolving compliance frameworks, supporting smoother commercialization pathways.
3. Rising Outsourcing of Complex Microbial Manufacturing
Building in-house microbiome production facilities requires substantial capital investment and specialized technical expertise. Consequently, biotechnology innovators increasingly rely on partnerships within the Live Biotherapeutic Products and Microbiome CDMO Market to:
- Reduce capital expenditure
- Accelerate time-to-market
- Access specialized fermentation technology
- Ensure regulatory compliance
Outsourcing strategies remain particularly attractive for emerging biotech firms pursuing asset-light business models.
4. Growing Pharmaceutical Interest in Microbiome Platforms
Large pharmaceutical companies are actively exploring microbiome collaborations to diversify biologics portfolios and strengthen precision medicine strategies.
Strategic partnerships, licensing agreements, and co-development models are reinforcing long-term demand within the Live Biotherapeutic Products and Microbiome CDMO Market, particularly as commercialization milestones approach.
Latest Blog :- https://www.researcheditorial.com/news/veterinary-microchips-market/
Emerging Trends Shaping the Live Biotherapeutic Products and Microbiome CDMO Market
Commercial-Scale Capacity Expansion
CDMOs are transitioning from pilot-scale production to commercial-scale infrastructure in anticipation of future regulatory approvals and market launches.
Dedicated Anaerobic Manufacturing Suites
Given the oxygen-sensitive nature of microbial strains, investment in high-containment anaerobic environments is increasing. Specialized fermentation suites are becoming core infrastructure within the Live Biotherapeutic Products and Microbiome CDMO Market.
Integrated Development and Manufacturing Models
Leading CDMOs are expanding service portfolios to include:
- Process optimization
- Formulation development
- Stability testing
- Regulatory documentation support
This integrated model strengthens long-term client retention and accelerates commercialization timelines.
Digitalization and Advanced Process Monitoring
Advanced analytics and real-time process monitoring technologies are being implemented to ensure:
- Strain consistency
- Contamination prevention
- Reproducibility at scale
Digital quality systems are emerging as a competitive differentiator in the Live Biotherapeutic Products and Microbiome CDMO Market.
Competitive Landscape: Specialization Defines Leadership
The Live Biotherapeutic Products and Microbiome CDMO Market is characterized by high technical specialization rather than commoditized pricing competition.
Key differentiators include:
- GMP-compliant anaerobic fermentation capabilities
- Microbial strain banking expertise
- Multi-strain formulation proficiency
- Scalable commercial production capacity
- Regulatory advisory support
Market participants are increasingly forming long-term strategic alliances, reflecting the lifecycle complexity of microbiome product development.
Regional Insights
North America
North America leads the Live Biotherapeutic Products and Microbiome CDMO Market due to:
- Advanced regulatory clarity
- Strong biotechnology investment ecosystem
- High concentration of microbiome-focused companies
- Established biologics manufacturing infrastructure
The United States remains the dominant regional contributor, while Canada continues to expand its innovation footprint.
Europe
Europe maintains a strong microbiome research base and regulatory framework, supporting steady CDMO demand across clinical and commercial stages.
Asia-Pacific
Asia-Pacific is emerging as a cost-competitive manufacturing region with growing biotechnology infrastructure. However, large-scale commercialization remains concentrated in Western markets.
Market Challenges
Despite robust growth prospects, the Live Biotherapeutic Products and Microbiome CDMO Market faces structural challenges:
- Strain viability and shelf-life limitations
- Contamination risks in multi-strain formulations
- Global regulatory harmonization
- Capital intensity of facility expansion
- Scaling complexities from clinical to commercial production
Strategic investment in infrastructure and quality systems will be essential to sustain growth momentum.
Future Outlook to 2035
The Global Live Biotherapeutic Products and Microbiome CDMO Market is projected to witness sustained high-growth expansion through 2035 as:
- Late-stage clinical approvals increase
- Physician adoption expands
- Commercial manufacturing demand accelerates
- Pharmaceutical partnerships intensify
The transition from early research to mainstream therapeutic commercialization represents a structural shift in biologics manufacturing.
CDMOs positioned with scalable anaerobic infrastructure and regulatory-aligned systems are expected to capture long-term competitive advantage within the Live Biotherapeutic Products and Microbiome CDMO Market.
Strategic Insights for Industry Stakeholders
Biotechnology Companies
- Engage CDMO partners early to de-risk scale-up.
- Prioritize regulatory-aligned microbial process development.
CDMO Executives
- Invest in dedicated microbiome production suites.
- Expand integrated development and compliance capabilities.
Pharmaceutical Leaders
- Evaluate microbiome collaborations to diversify biologics portfolios.
Investors
- Assess manufacturing readiness alongside clinical pipeline maturity when evaluating opportunities in the Live Biotherapeutic Products and Microbiome CDMO Market.
Conclusion
The Live Biotherapeutic Products and Microbiome CDMO Market represents one of the most dynamic frontiers in advanced biologics manufacturing. Scientific progress, regulatory maturity, and expanding commercialization pipelines are converging to create a sustained growth trajectory through 2035.
As microbiome therapeutics move into mainstream pharmaceutical adoption, scalable and compliant manufacturing will define long-term competitive positioning within the Live Biotherapeutic Products and Microbiome CDMO Market.
Organizations seeking actionable intelligence, competitive benchmarking, and forward-looking forecasting must adopt data-driven strategies to navigate this rapidly evolving ecosystem.
Read Full Report Here :- https://www.quintilereports.com/industry-report/1110-live-biotherapeutic-products-and-microbiome-cdmo-market/
Frequently Asked Questions (FAQs)
1. What is the Live Biotherapeutic Products and Microbiome CDMO Market?
The Live Biotherapeutic Products and Microbiome CDMO Market refers to the contract development and manufacturing ecosystem supporting pharmaceutical-grade microbiome-based therapies. It includes fermentation, formulation, GMP manufacturing, regulatory support, and commercialization services for live microbial therapeutics.
2. What are Live Biotherapeutic Products (LBPs)?
Live Biotherapeutic Products are regulated pharmaceutical formulations containing live microorganisms intended to prevent, treat, or manage diseases. Unlike dietary probiotics, LBPs undergo clinical trials, regulatory approval, and strict GMP-compliant manufacturing processes.
3. What is driving growth in the Microbiome CDMO Market?
Growth is primarily driven by expanding microbiome clinical pipelines, increasing regulatory clarity, rising outsourcing of complex biologics manufacturing, and growing pharmaceutical interest in microbiome-based precision therapies.
3. What is driving growth in the Microbiome CDMO Market?
Growth is primarily driven by expanding microbiome clinical pipelines, increasing regulatory clarity, rising outsourcing of complex biologics manufacturing, and growing pharmaceutical interest in microbiome-based precision therapies.
4. What is the projected market size by 2035?
The Live Biotherapeutic Products and Microbiome CDMO Market was valued at USD 78.05 million in 2026 and is projected to reach approximately USD 2,050.55 million by 2035, reflecting strong long-term expansion.
5. Why are pharmaceutical companies outsourcing microbiome manufacturing?
Microbiome manufacturing requires specialized anaerobic fermentation facilities, strain stability management, and strict contamination control. Outsourcing to specialized CDMOs reduces capital expenditure and accelerates commercialization timelines.
6. Which regions dominate the Live Biotherapeutic Products and Microbiome CDMO Market?
North America currently leads the market due to advanced regulatory frameworks, strong biotechnology investment, and established biologics manufacturing infrastructure. Europe and Asia-Pacific are also witnessing steady growth.
7. What are the key challenges in manufacturing live biotherapeutics?
Major challenges include microbial strain viability, shelf-life limitations, contamination risks in multi-strain formulations, regulatory harmonization across regions, and scaling production from clinical to commercial volumes.
7. What are the key challenges in manufacturing live biotherapeutics?
Major challenges include microbial strain viability, shelf-life limitations, contamination risks in multi-strain formulations, regulatory harmonization across regions, and scaling production from clinical to commercial volumes.
9. How are CDMOs differentiating themselves in this market?
CDMOs are differentiating through GMP-compliant anaerobic infrastructure, integrated development services, microbial strain banking expertise, regulatory advisory support, and scalable commercial production capacity.
10. What is the long-term outlook for the Microbiome CDMO Industry?
The market is expected to witness sustained high growth through 2035 as late-stage clinical approvals increase, physician adoption expands, and pharmaceutical partnerships accelerate global commercialization.